User:Mr. Ibrahem/Chloral hydrate
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Aquachloral, Novo-Chlorhydrate, Somnos, others |
| Other names | Trichloroacetaldehyde monohydrate |
| AHFS/Drugs.com | Monograph |
| Routes of administration | By mouth, rectal suppository |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Well absorbed |
| Metabolism | Liver and kidney (converted to trichloroethanol) |
| Elimination half-life | 8–10 hours |
| Excretion | Bile, feces, urine (various metabolites not unchanged) |
| Identifiers | |
| |
| Chemical and physical data | |
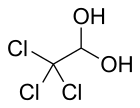
| Formula | C2H3Cl3O2 |
| Molar mass | 165.39 g·mol−1 |
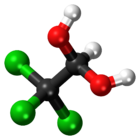
| 3D model (JSmol) | |
| Density | 1.9081 g/cm3 g/cm3 |
| Melting point | 57 °C (135 °F) |
| Boiling point | 98 °C (208 °F) |
| Solubility in water | 660 g/100 ml[1] |
| |
| |
Chloral hydrate has been used as a medication to treat trouble sleeping and in procedural sedation.[2][3] Use is only recommended when safer medications are not appropriate.[2] It can be taken by mouth or used rectally.[2] Onset of effects are within an hour and may last for up to eight hours.[2] Other uses include as a part of a stain for fungal spores.[4]
Common side effects include nausea, trouble with coordination, and diarrhea.[2] Other side effects may include addiction, delirium tremens if suddenly stopped, and allergic reactions.[2] Use is not recommended during pregnancy.[5] Excessive doses may result in decrease breathing or coma.[3] How it works in not entirely clear.[2]
Chloral hydrate was discovered in 1832 by Justus von Liebig and came into medical use in the 1860s.[6] It is available as a generic medication in the United Kingdom were 30 tablets costs the NHS about 140 pounds as of 2020.[5] It is not approved for any medical use in the United States, though is still occasionally used.[7][8] It remains commonly used in neonatal intensive care units in Europe.[9] Historically it was used in mental asylums to manage agitation and was mixed in alcohol to create the "knockout drops" known as a Mickey Finn.[10][11]
References[edit]
- ^ "Chemical Book: Chloral hydrate". Archived from the original on 29 August 2018. Retrieved 27 February 2017.
- ^ a b c d e f g h i "Chloral Hydrate Monograph for Professionals". Drugs.com. Archived from the original on 23 January 2021. Retrieved 12 December 2020.
- ^ a b c "Chloral Hydrate 143mg/5ml Oral Solution - Summary of Product Characteristics (SmPC) - (emc)". www.medicines.org.uk. Archived from the original on 29 August 2021. Retrieved 12 December 2020.
- ^ Auerbach, Paul S. (2011). Wilderness Medicine E-Book: Expert Consult Premium Edition - Enhanced Online Features. Elsevier Health Sciences. p. 1278. ISBN 978-1-4557-3356-9. Archived from the original on 2021-08-29. Retrieved 2020-12-12.
- ^ a b BNF 79. London: Pharmaceutical Press. March 2020. p. 502. ISBN 978-0857113658.
- ^ Stolerman, Ian (2010). Encyclopedia of Psychopharmacology. Springer Science & Business Media. p. 597. ISBN 978-3-540-68698-9. Archived from the original on 2021-08-29. Retrieved 2020-12-12.
- ^ Dowd, Frank J.; Johnson, Bart; Mariotti, Angelo (2016). Pharmacology and Therapeutics for Dentistry - E-Book. Elsevier Health Sciences. p. 167. ISBN 978-0-323-44595-5. Archived from the original on 2021-08-29. Retrieved 2020-12-12.
- ^ Meadows, Michelle (January–February 2007). "The FDA Takes Action Against Unapproved Drugs". FDA Consumer Magazine. 41 (1): 34–5. PMID 17342837. S2CID 37097870.
- ^ Lane, Robert H. (2014). Current Controversies in Perinatology, An Issue of Clinics in Perinatology, E-Book. Elsevier Health Sciences. p. 906. ISBN 978-0-323-32673-5. Archived from the original on 2021-08-29. Retrieved 2020-12-12.
- ^ "Chloral Hydrate". Drug Enforcement Administration. Archived from the original on 11 May 2012. Retrieved 27 June 2018.
- ^ Young, Mary de (2014). Madness: An American History of Mental Illness and Its Treatment. McFarland. p. 248. ISBN 978-0-7864-5746-5. Archived from the original on 2021-08-29. Retrieved 2020-12-12.
