User:Mr. Ibrahem/Nesiritide
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| Routes of administration | IV only |
| Drug class | Vasodilatation[1] |
| Chemical and physical data | |
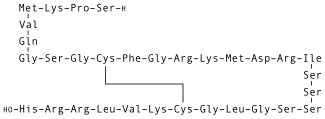
| Formula | C143H244N50O42S4 |
| Molar mass | 3464.07 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Nesiritide, sold under the brand name Natrecor among others, is a medication developed to treat acute decompensated congestive heart failure.[1] Evidence; however, has failed to show benefit.[2][1] It is given into a vein.[1]
Common side effects include low blood pressure, headache, nausea, and back pain.[1] Other side effects may include allergic reactions, kidney problems, and death.[1] It is a recombinant form of B-type natriuretic peptide, which results in vasodilatation.[1]
Nesiritide was approved for medical use in the United States in 2001.[1] In the United States it costs about 1,100 USD per 1.5 mg vial.[3] Commercial sale; however, has been discontinued as of 2021.[4]
References[edit]
- ^ a b c d e f g h i j "Nesiritide Monograph for Professionals". Drugs.com. Archived from the original on 22 June 2020. Retrieved 12 November 2021.
- ^ O'Connor CM, Starling RC, Hernandez AF, Armstrong PW, Dickstein K, Hasselblad V, et al. (July 2011). "Effect of nesiritide in patients with acute decompensated heart failure" (PDF). The New England Journal of Medicine. 365 (1): 32–43. doi:10.1056/NEJMoa1100171. hdl:11379/60663. PMID 21732835. Archived (PDF) from the original on 2021-02-11. Retrieved 2021-09-18.
- ^ "Natrecor Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 12 November 2021.
- ^ "Drugs@FDA: FDA-Approved Drugs". www.accessdata.fda.gov. Archived from the original on 21 March 2021. Retrieved 12 November 2021.
