User:Mr. Ibrahem/Dipyridamole
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Persantine, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682830 |
| Pregnancy category |
|
| Routes of administration | By mouth, IV |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 37–66%[1] |
| Protein binding | ~99% |
| Metabolism | Liver (glucuronidation)[2] |
| Elimination half-life | α phase: 40 min, β phase: 10 hours |
| Excretion | Biliary (95%), urine (negligible) |
| Identifiers | |
| |
| Chemical and physical data | |
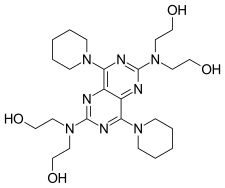
| Formula | C24H40N8O4 |
| Molar mass | 504.636 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Dipyridamole, sold under the brand name Persantine and others, is a medication used to prevent blood clots and as part of thallium myocardial perfusion imaging.[3] For blood clots prevention it is used together with either aspirin or warfarin.[4] It is taken by mouth or by injected into a vein.[3]
Side effects may include headache, dizziness, flushing, chest pain, gastrointestinal upset, nausea, diarrhea, rash, and itchiness.[4] Other side effects may include liver problems and anaphylaxis.[3] While there is no clear harm with use in pregnancy, such use has not been well studied.[5] It works by dilating blood vessels and decreasing platelet aggregation.[4]
Dipyridamole was approved for medical use in the United States in 1961.[3] It is available as a generic medication.[4] In the United Kingdom 60 pills of 200 mg costs the NHS about £13 as of 2021.[6] This amount in the United States costs about 88 USD.[7]
References[edit]
- ^ Nielsen-Kudsk, F; Pedersen, AK (May 1979). "Pharmacokinetics of Dipyridamole". Acta Pharmacologica et Toxicologica. 44 (5): 391–9. doi:10.1111/j.1600-0773.1979.tb02350.x. PMID 474151.
- ^ "Aggrenox (aspirin/extended-release dipyridamole) Capsules. Full Prescribing Information" (PDF). Boehringer Ingelheim Pharmaceuticals, Inc. Archived (PDF) from the original on 29 September 2016. Retrieved 1 December 2016.
- ^ a b c d e "Dipyridamole Monograph for Professionals". Drugs.com. Archived from the original on 12 May 2021. Retrieved 26 December 2021.
- ^ a b c d e "Dipyridamole". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 16 May 2021. Retrieved 26 December 2021.
- ^ "Dipyridamole Use During Pregnancy". Drugs.com. Archived from the original on 5 December 2020. Retrieved 26 December 2021.
- ^ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 135. ISBN 978-0857114105.
- ^ "Dipyridamole Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 26 December 2021.
