User:Mr. Ibrahem/Clofarabine
 | |
| Clinical data | |
|---|---|
| Trade names | Clolar, Evoltra, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607012 |
| License data |
|
| Routes of administration | Intravenous |
| Drug class | Antimetabolite[1] |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| Chemical and physical data | |
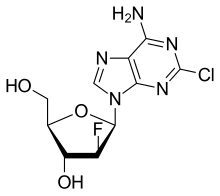
| Formula | C10H11ClFN5O3 |
| Molar mass | 303.68 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Clofarabine, sold under the brand name Clolar and Evoltra, is a medication used to treat acute lymphoblastic leukaemia (ALL).[3] It is used when other treatments stop working.[3] It is given by gradual injection into a vein.[3]
Common side effects include low white blood cells with a fever, anxiety, headache, flushing, nausea, diarrhea, itchiness, and tiredness.[1] Other side effects may include bone marrow suppression and tumor lysis syndrome.[3] Use during pregnancy may harm the baby.[3] It is a antimetabolite, specifically an analogue of adenine.[1] As such it disrupts DNA polymerase and RNA reductase preventing the making of new DNA and RNA.[1]
Clofarabine was approved for medical use in the United States in 2004 and Europe in 2006.[3][1] It is available as a generic medication.[4] In the United Kingdom 20 mg costs the NHS about £1,325 as of 2021.[4] In the United States this amount costs about 650 USD.[5]
References[edit]
- ^ a b c d e f g h "Evoltra". Archived from the original on 12 November 2020. Retrieved 5 January 2022.
- ^ "Clolar- clofarabine injection". DailyMed. 31 December 2019. Archived from the original on 13 August 2020. Retrieved 27 September 2020.
- ^ a b c d e f g "Clofarabine Monograph for Professionals". Drugs.com. Archived from the original on 16 August 2019. Retrieved 5 January 2022.
- ^ a b BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 953. ISBN 978-0857114105.
- ^ "Clofarabine Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 7 May 2016. Retrieved 5 January 2022.
