User:Mr. Ibrahem/Aprepitant
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Emend |
| Other names | Administer fosaprepitant dimeglumine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a604003 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth (capsules), intravenous |
| Drug class | Neurokinin-1 receptor antagonist[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 60–65% |
| Protein binding | >95% |
| Metabolism | Liver (mostly CYP3A4- mediated; some contributions by CYP2C19 & CYP1A2) |
| Elimination half-life | 9–13 hours |
| Excretion | Urine (5%), faeces (86%) |
| Identifiers | |
| |
| Chemical and physical data | |
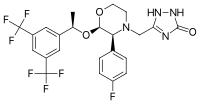
| Formula | C23H21F7N4O3 |
| Molar mass | 534.435 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Aprepitant, sold under the brand name Emend among others, is a medication used to prevent chemotherapy-induced nausea and vomiting (CINV) and to prevent postoperative nausea and vomiting.[1] It may be used together with ondansetron and dexamethasone.[1] It is taken by mouth.[1]
Common side effects include tiredness, loss of appetite, diarrhea, abdominal pain, hiccups, itchiness, pneumonia, and blood pressure changes.[1] Other severe side effects may include anaphylaxis.[1] While use in pregnancy does not appear to be harmful, such use has not been well studied.[3] Aprepitant belongs to the class of neurokinin-1 receptor antagonists medications.[1] It works by blocking substance P from attaching to the NK1 receptors.[4]
Aprepitant was approved for medical use in Europe and the United States in 2003.[1][4] It is made by Merck & Co.[1] It is on the World Health Organization's List of Essential Medicines.[5] In the United States it about US$230 per dose.[6] This amount in the United Kingdom cost the NHS about £16.[7] A form that can be given by injection into a vein, known as fosaprepitant is also available.[1]
References[edit]
- ^ a b c d e f g h i j k l "Aprepitant/Fosaprepitant Dimeglumine Monograph for Professionals". Drugs.com. Retrieved 13 October 2019.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Retrieved 11 September 2020.
- ^ "Aprepitant Use During Pregnancy". Drugs.com. Retrieved 13 October 2019.
- ^ a b "Emend". European Medicines Agency. 17 September 2018. Retrieved 13 October 2019.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Emend Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 13 October 2019.
- ^ "Aprepitant | Medicinal forms | BNFc content published by NICE". webcache.googleusercontent.com. NICE. Retrieved 13 October 2019.
